Norditropin SimpleXx 30iu (10mg/1,5 ml) Novo Nordisk description, Norditropin SimpleXx 30iu (10mg/1,5 ml) Novo Nordisk side effects, Norditropin SimpleXx 30iu (10mg/1,5 ml) Novo Nordisk price, Norditropin SimpleXx 30iu (10mg/1,5 ml) Novo Nordisk substance
Norditropin SimpleXx 5mg/1.5ml 15iu
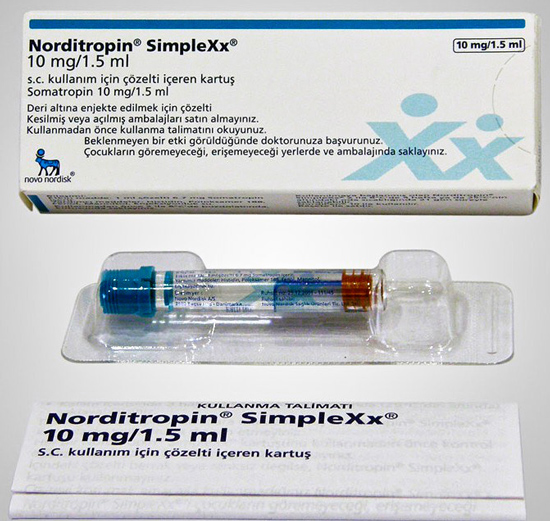
Norditropin SimpleXx 10mg/1.5ml 30iu
Norditropin SimpleXx 15mg/1.5ml 45iu
Norditropin SimpleXx 5 mg/1.5 mL, 10 mg/1.5 mL, 15 mg/1.5 mL;
Norditropin NordiFlex 5 mg/1.5 mL, 10 mg/1.5 mL, 15 mg/1.5 mL (SPC)
Manufacturer: Novo Nordisk
Pharmaceutical name: Human Growth Hormone 191aa
Pack: 1 cartridge (30 iu (10mg/1,5 ml))
Norditropin SimpleXx 10 mg/1.5 ml:
10 mg in a 1.5 ml of solution in a cartridge (Type I glass) closed at the bottom with a rubber stopper (Type I rubber closures) shaped as a plunger and at the top with a laminated rubber stopper (Type I rubber closures) shaped as a disc and sealed with an aluminium cap. The aluminium cap is sealed with a coloured cap (blue). Pack sizes of 1, 3 and 5.
Norditropin is a polypeptide hormone of recombinant DNA origin. The hormone is synthesized by a special strain of E. coli bacteria that has been modified by the addition of a plasmid which carries the gene for human growth hormone. Norditropin contains the identical sequence of 191 amino acids constituting the naturally occurring pituitary human growth hormone with a molecular weight of about 22,000 Daltons.
Therapy with Norditropin should be supervised by a physician who is experienced in the diagnosis and management of pediatric patients with short stature associated with GHD, Noonan syndrome, Turner syndrome or SGA, and adult patients with either childhood onset or adult onset GHD.
The Norditropin dosage and administration schedule should be individualized based on the growth response of each patient. Serum insulin-like growth factor I (IGF-I) levels may be useful during dose titration.
Norditropin injection is used to treat growth failure in children and adults who lack natural growth hormone, and in those with chronic kidney failure, Noonan syndrome, Turner syndrome, short stature at birth with no catch-up growth, and other causes. It is also used to prevent severe weight loss in people with AIDS, or to treat short bowel syndrome. It is a form of human growth hormone. Common side effects include headache, nausea, vomiting, fatigue, muscle pain, or weakness.
Norditropin may interact with insulin or oral diabetes medicine, steroids, cyclosporine, seizure medication, birth control pills, anabolic steroids, or hormone replacement medications for men or women. Tell your doctor all medications you use. Norditropin should be used only when prescribed during pregnancy. It is not known if this drug passes into breast milk. Consult your doctor before breast-feeding.
Norditropin SimpleXx 5mg/1.5ml (15IU) and 10mg (30IU)
Overview
Norditropin SimpleXx 5mg/1.5ml (15IU) and 10mg (30IU) solution for injection cartridges are indicated in children with growth problems, Turner’s syndrome, in people with certain kidney problems and in children who are too small for their age.
Norditropin is supplied by Novo Nordisk Limited and contains:
- histidine
- hydrochloric acid for pH adjustment
- mannitol
- phenol
- poloxamer 188
- sodium hydroxide for pH adjustment
- somatropin
- water for injections
Norditropin Simplexx is manufactured using recombinant DNA technology in E. coli secretion expression system.
Precautions
Caution must be taken if the patient:
- is a child who has stopped growing completely
- is over 60 years old
- is allergic or sensitive to any ingredients
- is suffering from complications caused by surgery, trauma, or other medical conditions
- has a respiratory infection, tumour, or kidney problems
- has diabetes or is at risk for developing diabetes e.g. family history of diabetes, has certain skin problems or is obese
- has or have had brain or central nervous system problems
- has pituitary problems
- has Prader-Willi syndrome
- has Silver-Russell syndrome
- has sleep apnoea
Norditropin SimpleXx is contraindicated in children under the age of four and in those who are having a kidney transplant.
Drug interaction
The following drugs may interact with Norditropin SimpleXx:
- ciclosporin
- gonadotrophin
- insulin
The following types of drugs may also interact with Norditropin SimpleXx:
- anabolic steroids
- anticonvulsants
- corticosteroids
- glucocorticosteroids
- hormones
- medicines that are metabolised by the cytochrome P450 system
- oestrogens
- sex steroids
- thyroid hormones
Doctor must be informed if the patients is taking any of the above, or is undergoing treatment with any alternative medicaments, including vitamins, herbal supplements, and non-prescription drugs.
Handling
For the best result from Norditropin SimpleXx it should be taken in the evening.
Do not use Norditropin SimpleXx if the packaging has been tampered with, or if it shows any signs of damage. If you started a cartridge, leave it in the pen. Once opened, store it in a fridge at 2-8°C, and dispose no later than 28 days after opening. You can also store it at the room temperature; however, you must dispose of it 21 days after opening. Store it in the carton box (it is light sensitive), out of the sight and reach of children and pets. Do not freeze, or use after the expiry date shown on the packaging.
Side-effects
In children, side-effects are usually uncommon or rare.
In adults, oedema of the extremities is very common (1 in 10). Less common side effects include headaches, joint pain or stiffness, muscle pain or tenderness and paraesthesia. In addition, carpal tunnel syndrome, diabetes, pain at the injection sites, itching or muscle stiffness is uncommon. On rare occasions skin rash may occur, sometimes in children.
Other symptoms (with unknown frequencies) include:
- abnormal laboratory test results
- bone problems
- ear problems especially in people with Turner's syndrome
- hypersensitivity reactions
- increase in the size of hands and feet
- increased blood glucose
- intracranial hypertension
- leukaemia
- metabolic problems
- production of anti-Norditropin SimpleXx antibodies
- reduced glucose tolerance
- thyroid problems
Norditropin Simplexx 15mg/1.5ml
Package leaflet: Information for the user
Norditropin�® SimpleXx�® 15 mg/1.5 ml solution for injection in cartridge
somatropin
Read all of this leaflet carefully before you start using this medicine because it contains important information for you.
- Keep this leaflet. You may need to read it again
- If you have any further questions, ask your doctor or pharmacist
- This medicine has been prescribed for you only. Do not pass it on to others. It may harm them, even if their signs of illness are the same as yours
- If you get any side effects talk to your doctor or pharmacist. This includes any possible side effects not listed in this leaflet. See section 4.
What is in this leaflet
1. What Norditropin�® SimpleXx�® is and what it is used for
2. What you need to know before you use Norditropin�® SimpleXx�®
3. How to use Norditropin�® SimpleXx�®
4. Possible side effects
5. How to store Norditropin�® SimpleXx�®
6. Contents of the pack and other information.
1. What Norditropin�® SimpleXx�® is and what it is used for
Norditropin�® SimpleXx�® contains a biosynthetic human growth hormone called somatropin which is identical to the growth hormone produced naturally in the body. Children need growth hormone to help them grow, but adults also need it for their general health.
Norditropin�® SimpleXx�® comes as a solution in a cartridge ready for you to inject after you have put it in the matching NordiPen�® injection pen.
Norditropin�® SimpleXx�® is used to treat growth failure in children:
- If they have no or very low production of growth hormone (growth hormone deficiency)
- If they have Turner syndrome (a genetic problem which may affect growth)
- If they have reduced kidney function
- If they are short and were born small for gestational age (SGA).
Norditropin�® SimpleXx�® is used as a growth hormone replacement in adults:
In adults Norditropin�® SimpleXx�® is used to replace growth hormone if their growth hormone production has been decreased since childhood or has been lost in adulthood because of a tumour, treatment of a tumour, or a disease that affects the gland which produces growth hormone. If you have been treated for growth hormone deficiency during childhood, you will be retested after completion of growth. If growth hormone deficiency is confirmed, you should continue treatment.
2. What you need to know before you use Norditropin�® SimpleXx�®
Do not use Norditropin�® SimpleXx�®
- If you are allergic to somatropin, to phenol, or to any of the other ingredients of this medicine (listed in section 6)
- If you have had a kidney transplant
- If you have an active tumour (cancer). Tumours must be inactive and you must have finished your anti-tumour treatment before you start your treatment with Norditropin�® SimpleXx�®
- If you have an acute critical illness e.g. open heart surgery, abdominal surgery, multiple accidental trauma, or acute respiratory failure
- If you have stopped growing (closed epiphyses) and you do not have growth hormone deficiency.
Warnings and precautions
Talk to you doctor or pharmacist before using Norditropin�® SimpleXx�®
- If you have diabetes
- If you have ever had a cancer or another kind of tumour
- If you have recurrent headaches, eyesight problems, nausea, or if vomiting occurs
- If you have abnormal thyroid function
- If you develop a limp or low-back pain as these could be signs of a curved spine (scoliosis)
- If you are over 60 years of age, or have received somatropin treatment as an adult for more than 5 years, as experience is limited
- If you suffer from kidney disease as your kidney function should be monitored by your physician.
Other medicines and Norditropin�® SimpleXx�®
Tell your doctor or pharmacist if you are using, have recently used or might use any other medicines
- Glucocorticoids or sex steroids (for example anabolic steroids and estrogen) — your adult height may be affected if you use Norditropin�® SimpleXx�® and glucocorticoids or sex steroids at the same time
- Cyclosporin (immunosuppressive) — as your dose may need to be adjusted
- Insulin — as your dose may need to be adjusted
- Thyroid hormone — as your dose may need to be adjusted
- Gonadotrophin (gonad stimulating hormone) — as your dose may need to be adjusted
- Anticonvulsants — as your dose may need to be adjusted.
Pregnancy and breast-feeding
Somatropin containing products are not recommended in women of childbearing potential not using contraception.
- Pregnancy. Stop the treatment and tell your doctor if you become pregnant while you are using Norditropin�® SimpleXx�®
- Breast-feeding. Do not use Norditropin�® SimpleXx�® while you are breast-feeding as somatropin might pass into your milk.
Driving and using machines
Norditropin�® SimpleXx�® does not affect the use of any machines or the ability to drive safely.
3. How to use Norditropin�® SimpleXx�®
Always use this medicine exactly as your doctor has told you. Check with your doctor or pharmacist if you are not sure.
Recommended dose
The dose for children depends on their body weight and body surface area. Later in life, the dose depends on height, weight, gender and growth hormone sensitivity and will be adjusted until you are on the right dose.
- Children with low production or lack of growth hormone: The usual dose is 0.025 to 0.035 mg per kg body weight per day or 0.7 to 1.0 mg per m2 body surface area per day
- Children with Turner syndrome: The usual dose is 0.045 to 0.067 mg per kg body weight per day or 1.3 to 2.0 mg per m2 body surface area per day
- Children with kidney disease: The usual dose is 0.05 mg per kg body weight per day or 1.4 mg per m2 body surface area per day
- Children born small for gestational age (SGA): The usual dose is 0.035 mg per kg body weight per day or 1.0 mg per m2 body surface area per day until final height is reached. (In clinical trials of short children born SGA doses of 0.033 and 0.067 mg per kg body weight per day have typically been used)
- Adults with low production or lack of growth hormone: If your growth hormone deficiency continues after completion of growth, treatment should be continued. The usual starting dose is 0.2 to 0.5 mg per day. Dose will be adjusted until you are on the right dose. If your growth hormone deficiency starts during adult life, the usual starting dose is 0.1 to 0.3 mg per day. Your doctor will increase this dose each month until you are on the right dose. The usual maximum dose is 1.0 mg per day.
When to use Norditropin�® SimpleXx�®
- Inject your daily dose into the skin every evening just before bedtime.
How to use Norditropin�® SimpleXx�®
- Norditropin�® SimpleXx�® solution comes in cartridges with a color coded cap and is ready to be used in the matching color coded NordiPen�®�®. If the corresponding NordiPen�®�® is not used it will result in incorrect dosing. The NordiPen�® instruction manual tells you how to use the cartridges in the injection pen
- Check each new Norditropin�® SimpleXx�® cartridge before you use it. Do not use any cartridge that is damaged or cracked
- Do not use the Norditropin�® SimpleXx�® cartridges if the solution inside is cloudy or discoloured
- Vary the area you inject so you do not harm your skin
- Do not share your Norditropin�® SimpleXx�® cartridge with anyone else.
How long you will need treatment for
- Children with growth failure because of Turner syndrome, kidney disease, or SGA: Your doctor will recommend you continue treatment until you stop growing
- Children or adolescents who lack growth hormone: Your doctor will recommend you continue treatment into adulthood
- Do not stop using Norditropin�® SimpleXx�® without discussing it with your doctor first.
If you use more Norditropin�® SimpleXx�® than you should:
Tell your doctor if you inject too much somatropin. Long-term overdosing can cause abnormal growth and coarsening of facial features.
If you forget to use Norditropin�® SimpleXx�®:
Take the next dose as usual, at the normal time. Do not take a double dose to make up for a forgotten dose.
If you stop using Norditropin�® SimpleXx�®
Do not stop using Norditropin�® SimpleXx�® without discussing it with your doctor first.
If you have any further questions on the use of this medicine, ask your doctor or pharmacist.
4. Possible side effects
Like all medicines, this medicine can cause side effects, although not everybody gets them.
Effects seen in children and adults (unknown frequency):
- Rash; wheezing; swollen eyelids, face or lips; complete collapse. Any of these may be signs of allergic reactions
- Headache, eyesight problems, feeling sick (nausea), and being sick (vomiting). These may be signs of raised pressure in the brain
- Serum thyroxin levels may decrease
- Hyperglycaemia (elevated levels of blood glucose).
If you get any of these effects, see a doctor as soon as possible. Stop using Norditropin�® SimpleXx�® until your doctor says you can continue treatment.
Formation of antibodies directed against somatropin has been rarely observed during Norditropin�® therapy.
Increased levels of liver enzymes have been reported.
Cases of leukaemia and relapse of brain tumours have also been reported in patients treated with somatropin (the active ingredient in Norditropin�® SimpleXx�®), although there is no evidence that somatropin was responsible.
If you think you are suffering from any of these diseases, speak to your doctor.
Additional side effects in children:
Uncommon (may affect up to 1 in 100 children):
- Headache
- Redness, itching, and pain in the area of injection.
Rare (may affect up to 1 in 1,000 children):
- Rash
- Muscle and joint pain
- Swollen hands and feet due to fluid retention.
In rare cases, children using Norditropin�® SimpleXx�® have experienced hip and knee pain or have started limping. These symptoms may be caused by a disease affecting the top of the thigh bone (Legg-Calve disease) or because the end of the bone has slipped from the cartilage (slipped capital femoral epiphysis) and may not be due to Norditropin�® SimpleXx�®.
In children with Turner syndrome, a few cases of increased growth of hands and feet compared to height have been observed in clinical trials.
A clinical trial in children with Turner syndrome has shown that high doses of Norditropin�® can possibly increase the risk of getting ear infections.
If any of these side effects gets serious, or if you notice any side effects not listed in this leaflet, please tell your doctor or pharmacist, as the dose may need to be reduced.
Additional side effects in adults:
Very common (may affect more than 1 in 10 adults):
- Swollen hands and feet due to fluid retention.
Common (may affect up to 1 in 10 adults):
- Headache
- Feeling of skin crawling (formication) and numbness or pain mainly in fingers
- Joint pain and stiffness; muscle pain.
Uncommon (may affect up to 1 in 100 adults):
- Type 2 diabetes
- Carpal tunnel syndrome; tingling and pain in fingers and hands
- Itching (can be intense) and pain in the area of injection
- Muscle stiffness.
Reporting of side effects
If you get any side effect, talk to your doctor or pharmacist. This includes any possible side effects not listed in this leaflet.
You can also report side effects directly via the Yellow Card Scheme Website: www.mhra.gov.uk/yellowcard. By reporting side effects you can help provide more information on the safety of this medicine.
5. How to store Norditropin�® SimpleXx�®
Keep this medicine out of the sight and reach of children.
Do not use this medicine after the expiry date which is stated on the package after EXP/. The expiry date refers to the last day of that month.
Store unused Norditropin�® SimpleXx�® cartridges in a refrigerator (2°C — 8°C) in the outer carton, in order to protect from light. Do not freeze or expose to heat.
While using Norditropin�® SimpleXx�® 15 mg/1.5 ml cartridge in an injection pen you can either:
- Keep it in the pen for up to 4 weeks in a refrigerator (2°C — 8°C), or
- Keep it in the pen for up to 3 weeks at room temperature (below 25°C).
Do not continue to use Norditropin�® SimpleXx�® cartridges if they have been frozen or exposed to excessive temperatures.
Check each new Norditropin�® SimpleXx�® cartridge before you use it. Do not use any cartridge that is damaged or cracked.
Do not use Norditropin�® SimpleXx�® cartridges if the solution inside is cloudy or discoloured.
Do not throw away any medicines via wastewater or household waste. Ask your pharmacist how to throw away medicines you no longer use. These measures will help protect the environment.
6. Contents of the pack and other information
What Norditropin�® SimpleXx�® contains
- The active substance is somatropin
- The other excipients are mannitol; histidine; poloxamer 188; phenol; water for injection; hydrochloric acid; and sodium hydroxide.
What Norditropin�® SimpleXx�® looks like and contents of the pack
Norditropin�® SimpleXx�® is a clear and colourless solution in a 1.5 ml glass cartridge ready for injection in the NordiPen�® injection pen.
1 ml of solution contains 10 mg somatropin.
1 mg of somatropin corresponds to 3 IU of somatropin.
Norditropin�® SimpleXx�® is available in three strengths:
5 mg/1.5 ml, 10 mg/1.5 ml and 15 mg/1.5 ml (equivalent to 3.3 mg/ml, 6.7 mg/ml and 10 mg/ml, respectively).
Marketing Authorisation Holder and Manufacturer
Marketing Authorisation Holder
Novo Nordisk Limited
3 City Place
Beehive Ring Road
Gatwick
West Sussex
RH6 0PA
Manufacturer
Novo Nordisk A/S
Novo Alle
DK-2880 Bagsv?rd
Denmark
This medicinal product is authorised in the Member States of the EEA under the following names:
Austria, Belgium, Croatia, Cyprus, Czech Republic, Denmark, Finland, Germany, Greece, Hungary, Ireland, Italy, Luxembourg, Netherlands, Portugal, Spain, Slovenia, Sweden, United Kingdom: Norditropin�® SimpleXx�® 15 mg/1.5 ml
France: Norditropine�® SimpleXx�® 15 mg/1.5 ml
This leaflet was last revised in 05/2015
Norditropin�®, SimpleXx�® and NordiPen�® are trademarks owned by Novo Nordisk Health Care AG, Switzerland
Novo Nordisk A/S
